When a patent dies for inequitable conduct, the damage goes far beyond losing a single lawsuit. The entire asset vanishes: the injunction disappears, licensing revenue collapses mid-quarter, and the victorious defendant often pivots to a Walker Process antitrust counterclaim seeking treble damages and attorney fees. This is the quiet, catastrophic risk lurking behind every prosecution file.
That risk stems from a strict, personal obligation: 37 CFR 1.56 imposes a duty of candor on every inventor, attorney, and anyone substantively involved in prosecution to disclose all information “material to patentability” to the USPTO. Under the post-Therasense but-for standard, information is material if a claim would not have issued had the examiner known about it. The duty reaches prior art, foreign office actions from counterpart applications, and statements made to other regulators — including the FDA and EMA — that contradict positions taken at the USPTO.
Key Takeaways
- The 37 CFR 1.56 rule is non-delegable. Disclosure obligations cannot be pushed onto outside counsel or assumed to be “someone else’s job.”
- The gap between regulatory and judicial materiality is where the dangerous compliance errors live. Post-Therasense, litigation applies a narrow but-for standard while the USPTO’s regulatory standard under 37 CFR 1.56(b) remains broader. The safer posture in high-value families is to disclose against the broader standard.
- A proven violation of 37 CFR 1.56 renders the patent permanently unenforceable, can infect related patents in the same family, and can open the door to Walker Process antitrust counterclaims with treble damages under § 4 of the Clayton Act.
- Foreign counterpart applications and cross-agency regulatory statements are the two most overlooked exposure points of inequitable conduct under 37 CFR 1.56.
- A documented 37 CFR 1.56 compliance program is the most effective rebuttal to specific-intent allegations. Contemporaneous records, centralized prior-art tracking, foreign-communication docketing protocols, and cross-functional IP/regulatory review make deceptive intent factually implausible on the record.
The Legal Foundation: What 37 CFR 1.56 Requires and Who It Binds
37 CFR 1.56 requires full disclosure of all known information that could affect patentability, not only from inventors and attorneys, but anyone who reviews, drafts, or influences what goes to the USPTO. While the USPTO outlines the rule in MPEP § 2001.01 et seq., the definition that truly matters emerges in litigation. When inequitable conduct is pleaded in an infringement case, federal courts apply the post-Therasense but-for test to decide materiality, and that judicial standard is narrower than what the prosecution context implies.
Decoding the Materiality Standard: Why the Post-Therasense But-For Test Redefined Disclosure Strategy
In Therasense, Inc. v. Becton, Dickinson & Co., 649 F.3d 1276 (Fed. Cir. 2011) (en banc), the Federal Circuit did something rare: it rewrote its own doctrine. For nearly three decades courts had applied a broad “reasonable examiner” test — information was material if a reasonable examiner would have considered it important. That standard, combined with a sliding scale that let strong materiality rescue weak intent, turned the inequitable conduct defense into what the Federal Circuit in Therasense called a “plague” on the patent system.
Therasense killed the “sliding scale”. It replaced the reasonable-examiner test with a but-for standard: information is but-for material only if the USPTO would not have allowed a claim had it been aware of the undisclosed reference. Materiality and intent must each be proved independently, by clear and convincing evidence. Negligence is not enough. Gross negligence is not enough. The accused infringer must show that specific intent to deceive is “the single most reasonable inference” that can be drawn from the evidence.
One carve-out survived. The Therasense court preserved an “affirmative egregious misconduct” exception that operates independently of the but-for test. A knowingly false affidavit, a fabricated inventor declaration, or a deliberate misrepresentation about the state of the prior art can trigger unenforceability even if the misconduct would not satisfy but-for materiality on its own. The exception is narrow. But it is the reason no practitioner should ever treat “well, the claim would have issued anyway” as a complete defense.
Here is a compliance blind spot that practitioners often miss. 37 CFR 1.56 — the regulatory standard the USPTO applies during prosecution — remains broader than the judicial but-for standard applied in litigation. The PTO’s rule still treats information as material if it establishes a prima facie case of unpatentability or is inconsistent with a position taken during prosecution. That means a reference can be undisclosed without triggering judicial unenforceability under Therasense, yet still expose the practitioner to PTO discipline. The more dangerous scenario runs the other way: a practitioner relies on the narrower judicial standard to justify withholding, and a litigation opponent later reconstructs the prosecution file and argues — successfully — that but-for materiality was satisfied all along. The safer assumption in any high-value patent family is that the broader regulatory standard governs the disclosure decision, and the narrower judicial standard is what a defendant will try to meet with the benefit of hindsight.
Consider how this plays out in practice. An applicant prosecutes a claim to compound X, arguing a 30% efficacy improvement over the closest prior art. During prosecution, a foreign associate forwards an EPO search report citing a structurally similar compound that achieved 28% improvement — at a different dosage, in a different patient population. U.S. counsel concludes the reference is distinguishable and declines to submit it on the IDS. Five years later, in a district court in Delaware, the accused infringer’s expert testifies that at equivalent dosages the prior art compound would have shown 31% improvement, enough to anticipate the claim. Under the broader regulatory standard, the reference was plainly material. Under the but-for standard, materiality now turns on an expert battle the patent holder may lose. The decision to withhold, made in a minute during prosecution, becomes a seven-figure problem in litigation.
Identifying Every Individual Bound by the Duty — and Why It’s Personal and Non-Delegable
The rule binds a wider universe than most in-house teams assume. 37 CFR 1.56(c) reaches every inventor named in the application, every attorney or agent who prepares or prosecutes it, and every other person substantively involved in its preparation or prosecution who is associated with the inventor, the applicant, or an assignee. The duty is personal. It cannot be delegated to outside counsel. It is not discharged when a foreign associate forwards a search report to one attorney in the chain.
For an in-house IP strategist, this creates a cascade problem that most compliance programs understate. Picture the actual fact pattern: a foreign associate in Munich forwards an EPO search report, and the email has six people on the CC line — two in-house patent attorneys, a senior inventor, a technical director, an outside prosecution partner, and the VP of regulatory affairs. At the moment that email lands, each of those six people has just acquired an independent, personal disclosure obligation with respect to any material reference it contains. Six personal duties, from one forwarded email — and every one of them is reconstructible in discovery years later. Courts have consistently rejected the defense that an individual reasonably assumed someone else in the chain would handle it. The duty does not work that way.
The knowledge threshold is actual awareness, not constructive knowledge. In theory, this is protective: a practitioner who genuinely did not know of a reference cannot be charged with failing to disclose it. In practice, the protection is thinner than it reads. Email threads, docketing entries, deposition transcripts, and internal memoranda are routinely used to establish what an individual knew and when. Arguments that a reference was “technically in the file but no one read it” have a long history of failing in front of Federal Circuit panels, because the inference courts are willing to draw against sophisticated corporate actors runs against the patentee, not in its favor.
With the rule and its reach defined, the operational question becomes: what does compliance look like on the page? That is the job of the Information Disclosure Statement.
The Information Disclosure Statement: Your Primary Compliance Weapon
The Information Disclosure Statement (IDS) is the most visible compliance artifact in any prosecution record. When litigators build an inequitable conduct defense, they start by reconstructing what was disclosed, when, and by whom; gaps in this record are where narratives of deceptive intent are assembled. A well-constructed IDS does the opposite: it documents diligence, signals good faith, and narrows the factual territory a litigation opponent can exploit. Understanding what must go into the IDS is deceptively simple on the surface—patents, publications, and other information known to be material to patentability—but the “long list” that actually drives inequitable conduct litigation reaches much further. For a complete guide on the rules and procedures, see everything you need to know about filing an IDS.
The duty to disclose does not end at issuance; it runs through ex parte reexamination, inter partes review, post-grant review, and every continuation or divisional in the same family. Each new procedural posture can reset the compliance clock in ways that catch teams off guard, making mastering IDS timing critical. Prior art surfaced during litigation discovery is a classic trap: a reference produced by an accused infringer in document discovery or cited in an IPR petition is immediately known to the patent holder’s team. If there is a co-pending continuation, reexamination, or related application anywhere in the portfolio, that reference must be evaluated for disclosure immediately—not at the next scheduled filing. Failure to disclose material information surfaced during parallel proceedings is an independent basis for an inequitable conduct finding, one courts treat as especially damning because the knowledge trail is pristine.
The IDS is the tool, but the stakes of misusing it—or failing to use it—belong to a different vocabulary: inequitable conduct, the so-called “death penalty” of patent law.
Inequitable Conduct: The Death Penalty for 37 CFR 1.56 Violations
Call it what it is. A patent found unenforceable for inequitable conduct is permanently unenforceable. No cure. No amendment. No reissue saves it. The Federal Circuit has called the remedy the “atomic bomb” of patent law, and the label is accurate in two dimensions: it destroys the target patent, and the blast radius can reach every related application in the family.
The Two-Prong Test: Proving Materiality and Specific Intent to Deceive
Post-Therasense, an accused infringer pleading inequitable conduct must prove two elements, each by clear and convincing evidence: (a) but-for materiality of the withheld information or misrepresentation, and (b) specific intent to deceive the USPTO. The two prongs operate independently. A strong materiality showing cannot rescue a weak intent case, or the reverse — the sliding scale is gone.
The intent prong is where most post-Therasense cases are decided. Direct evidence of deceptive intent is almost nonexistent — no rational practitioner memorializes a decision to defraud the USPTO — so courts infer intent from circumstantial evidence. Therasense imposed a demanding version of that inference. Specific intent to deceive must be “the single most reasonable inference” drawn from the evidence. Where multiple reasonable inferences remain on the table — negligence, misunderstanding, a good-faith judgment call — the intent prong fails.
This standard does more work than it reads on the page. A litigator building an inequitable conduct narrative will reconstruct the prosecution timeline with forensic care: when a reference arrived, who received it, what was said about it internally, why it was not submitted. The goal is to eliminate every plausible explanation other than deceptive intent. A contemporaneous record that documents good-faith disclosure decisions — even imperfect ones — is usually what defeats the “single most reasonable inference” argument. A prosecution file showing a reference arriving, a docketing entry, an internal review, and a reasoned (if ultimately wrong) conclusion not to disclose is a completely different file from one that shows a reference arriving and then nothing.
Consider the contrast. An EPO search report arrives citing a reference. The docketing system logs the receipt. A U.S. associate reviews the reference, concludes it is cumulative over what the examiner already has, and memorializes that judgment in a two-paragraph internal note. Five years later a district court disagrees about cumulativeness. But because multiple reasonable inferences remain — the associate could have been right, could have been negligent, could have made a good-faith judgment call — the intent prong fails. Now the same reference, different file. The reference arrives. No docketing entry. No internal note. The associate who received the email has since left the company and cannot be deposed. The vacuum itself is where the “single most reasonable inference” of deceptive intent gets constructed.
Infectious Unenforceability: How One Patent’s Misconduct Can Contaminate an Entire Portfolio
The remedy for inequitable conduct does not stay in its lane. Under the doctrine of infectious unenforceability, courts can extend an unenforceability ruling to related patents that share an immediate and necessary relation to the tainted prosecution. For a portfolio built around a core technology with continuations, divisionals, and parallel prosecution streams, this is the scenario that keeps chief patent counsel awake.
Courts look to several factual triggers when deciding whether to extend unenforceability: shared inventors between the tainted patent and the related patents; shared prosecution counsel who had access to the same withheld information; overlapping claim scope or claim language derived from the same disclosure; and continuation strategies that appear designed to obscure or relitigate the same prior art. No single factor automatically extends unenforceability. Together, they create the factual predicate courts use to do so.
The business consequences scale accordingly. A single inequitable conduct finding in a flagship patent can eliminate injunctive relief across the entire family, collapse licensing revenue streams built on the portfolio’s enforceability, and — as discussed below — open the door to Walker Process antitrust exposure. For pharma portfolios built around composition-of-matter patents and method-of-use continuations, infectious unenforceability can end a product’s market exclusivity years before nominal patent expiry.
The strategic response is structural. Prosecution teams for high-value families should be designed to contain the blast radius: rotate prosecution counsel across related applications where possible, document information-sharing protocols between teams, and maintain parallel but independent disclosure records for each application so that a finding about one prosecution does not automatically pull in the others. For portfolios already in place, a pre-litigation audit can identify the shared-counsel, shared-inventor, and overlapping-scope factors a future defendant will use to argue for family-wide unenforceability. That is the work patent portfolio analysis performs before a litigation opponent does it on their own timeline.
If this risk mapping describes your portfolio, it may be time to audit it before a litigation opponent does.
Fraud vs. Inequitable Conduct vs. Walker Process — How the Three Doctrines Differ in Litigation
Three related but distinct doctrines sit in this territory, and conflating them is how practitioners lose control of the risk conversation. Each has its own elements, its own proof standard, and its own remedy.
| Doctrine | Elements | Proof Standard | Remedy |
| Inequitable Conduct | (1) But-for materiality of withheld information (or affirmative egregious misconduct) + (2) specific intent to deceive the USPTO | Clear and convincing evidence | Patent rendered permanently unenforceable; potential infectious unenforceability of related patents; exceptional case finding supporting attorney fees |
| Common-Law Fraud on the USPTO | (1) Material misrepresentation + (2) knowledge of falsity + (3) intent to induce reliance + (4) actual reliance by the USPTO + (5) resulting damages | Clear and convincing evidence | Unenforceability of the patent; damages requires separate proof |
| Walker Process Fraud (Walker Process Equipment, Inc. v. Food Machinery & Chemical Corp., 382 U.S. 172 (1965)) | (1) Knowing and willful fraud on the USPTO (higher than inequitable conduct — specific intent plus reliance) + (2) enforcement of the fraudulently procured patent + (3) relevant market definition + (4) monopoly power or dangerous probability thereof + (5) antitrust injury | Clear and convincing evidence as to the fraud element; preponderance as to antitrust elements | Treble damages under § 4 of the Clayton Act; attorney fees; injunctive relief under the antitrust laws |
The practical distinctions matter. Inequitable conduct is cheaper for the defendant to prove and harder for the patentee to escape — no reliance or damages required, and the remedy attaches automatically on a finding. Common-law fraud requires the full tort elements and is rarely litigated as a standalone theory. Walker Process sits at the top of the pyramid: the hardest standard to meet, because it requires knowing-and-willful fraud plus independent proof of antitrust injury and market power — but it carries the worst downside. Treble damages on top of attorney fees, applied against the revenue extracted during enforcement, can turn an inequitable conduct case into a company-ending event.
The pathway from inequitable conduct to Walker Process is the scenario to understand. An inequitable conduct finding is not automatically a Walker Process violation — the standards differ, and the Federal Circuit has been clear that Walker Process requires the higher knowing-and-willful fraud showing. But inequitable conduct findings create the factual record from which Walker Process counterclaims are built. The most dangerous pathway involves statements made during prosecution that are later contradicted by internal company documents discovered in litigation. When a prosecution declaration asserts one set of facts and an internal email from the same period asserts the opposite, the two-document pattern is what transforms an inequitable conduct defense into a Walker Process counterclaim — and the company learns, far too late, that its patent file and its internal files were telling inconsistent stories the entire time.
These consequences are severe enough when prosecution stays in a single jurisdiction. In a global filing strategy, the attack surface multiplies because every foreign counterpart is a potential source of undisclosed material information.
Global Filing Strategy: The Cross-Border Disclosure Obligations That Quietly Destroy Patents
Here is the operational reality most inequitable conduct findings in the past decade trace back to: cross-border workflow failure, not doctrinal ambiguity. The law is actually clearer at the border than in many domestic scenarios. What fails is the plumbing between U.S. prosecution counsel, foreign associates, and — in pharma and biotech — the regulatory affairs function preparing FDA and EMA submissions on the same technology the patent team is prosecuting.
Foreign Counterpart Applications: The Most Dangerous Disclosure Gap in Modern Patent Prosecution
The obligation is specific. Prior art cited in a foreign counterpart application, rejections issued by foreign patent offices, and search reports generated in foreign or PCT proceedings must all be evaluated for U.S. disclosure. That reaches PCT International Search Reports, European Patent Office communications — including written opinions and examination reports — national phase office actions from every jurisdiction, and prior art surfaced in EPO opposition proceedings.
The case that defined the risk is Therasense itself. Abbott’s inequitable conduct problem was not a missed piece of prior art. It was a set of briefs Abbott had filed at the European Patent Office — about a counterpart patent — stating that a protective membrane was “unequivocally optional,” while simultaneously telling the USPTO that a person skilled in the art would understand the same protective membrane to be effectively required for whole blood testing. The EPO briefs were not disclosed to the USPTO. When a U.S. district court compared the two positions and found them irreconcilable, the patent was held unenforceable. A Federal Circuit en banc opinion later revised the doctrinal framework. The core diagnosis — that inconsistent positions across patent offices will end a patent — survived the rewrite.
The workflow breakdown that produces these outcomes is predictable. Foreign associates file responses to foreign office actions without systematically notifying U.S. prosecution counsel, either because the engagement was not structured to require it or because the operating procedure was “tell U.S. counsel if you think something is important.” U.S. prosecution counsel does not implement docketing triggers that capture foreign search reports, EPO communications, or national phase office actions on a defined timeline. Opposition proceedings at the EPO — where third parties submit prior art and the board of appeal issues reasoned opinions — surface references that never make it back across the Atlantic. Each of these gaps is survivable on its own. Stacked together, they produce a prosecution record where material information was available to someone on the patent holder’s team but was never evaluated for U.S. disclosure by anyone.
The fix is operational, not doctrinal. The protocol below is a minimum viable workflow that closes the most common gaps.
From Foreign Office Action to U.S. IDS Filing — The 7-Step Protocol:
- Receipt trigger. Every foreign communication — office action, search report, EPO written opinion, national phase rejection, opposition submission — is docketed at the point of receipt by the foreign associate, with an automatic timestamp and a copy to a designated U.S. counsel address.
- 24-hour acknowledgment. U.S. counsel acknowledges receipt within 24 business hours. This is a forensic step: it fixes the knowledge date for purposes of the 1.97(e) three-month certification clock.
- Translation initiation (where required). Non-English communications are routed for translation within 3 business days. Translation delay is not a legal excuse for disclosure delay if the reference is substantively material.
- Materiality screening. A designated U.S. attorney reviews each cited reference against the claims of every co-pending U.S. application in the same family. The screening decision — disclose, do not disclose, flag for further review — is documented contemporaneously.
- Cross-application check. References flagged for disclosure are checked against every related application in the portfolio, not just the specific counterpart. Continuations, divisionals, and parallel prosecutions in the same family trigger independent disclosure obligations.
- IDS preparation and filing. An IDS is prepared and filed in every relevant application within the window that preserves the lowest-risk certification posture — ideally within 3 months of the foreign-communication trigger date to preserve 1.97(e)(1) eligibility.
- Audit log. The complete sequence — receipt, acknowledgment, translation, screening decision, cross-application check, IDS filing — is preserved in a defensible audit log producible in response to a future inequitable conduct discovery request.
This protocol will not prevent every inequitable conduct allegation. It makes most of them impossible to win.
Inconsistent Statements Across Regulatory Agencies: The Pharma and Biotech Flashpoint
For pharma and biotech portfolios, the duty of candor reaches into workflows most companies do not realize are connected to patent prosecution at all. Statements made to the FDA during an NDA or BLA submission, statements made to the EMA in a marketing authorization application, and statements made in post-approval pharmacovigilance reporting can all become inequitable conduct evidence if they contradict positions taken at the USPTO.
Aventis Pharma S.A. v. Amphastar Pharmaceuticals, Inc., 525 F.3d 1334 (Fed. Cir. 2008), is the cautionary case. Aventis prosecuted a patent covering a low-molecular-weight heparin marketed as Lovenox and overcame prior art rejections by submitting declarations comparing the patented compound’s half-life to a prior art European compound. The half-life numbers looked impressive. The examiner allowed the claims. What Aventis did not tell the USPTO was that the half-life data had been generated by comparing a 40 mg dose of the patented compound against a 60 mg dose of the prior art. At equivalent doses, the half-life difference was not statistically significant. Amphastar reconstructed this from the underlying data in litigation. The Federal Circuit affirmed a finding of inequitable conduct, and the Lovenox patent was held unenforceable. Aventis’s exclusivity on a blockbuster anticoagulant ended years before the patent would have expired.
The Aventis pattern is not unique. FDA submission databases, FOIA-produced correspondence, and post-approval safety reports are all discoverable in patent litigation. When a drug’s mechanism of action is described one way to the FDA and another way to the USPTO, or when the prior-art landscape characterized to the FDA differs from the one characterized to the examiner, the inconsistency is reconstructible — and generic challenger counsel is highly motivated to reconstruct it.
The cross-functional compliance response requires organizational design, not just better legal review. IP counsel should be integrated into the review cycle for regulatory submissions that discuss mechanism of action, comparative efficacy, and prior-art landscape — precisely the categories of regulatory statements that most commonly surface in later patent litigation. The reverse is equally true. Regulatory affairs should be notified when patent prosecution arguments rely on representations about a drug’s properties, so that subsequent FDA correspondence does not inadvertently contradict the prosecution record. This is the protocol most portfolios do not have, and its absence is what turns a successful FDA submission into an inequitable conduct exhibit five years later.
Understanding the risk is half the battle. The other half is operationalizing defense — which is where compliance infrastructure becomes the real differentiator.
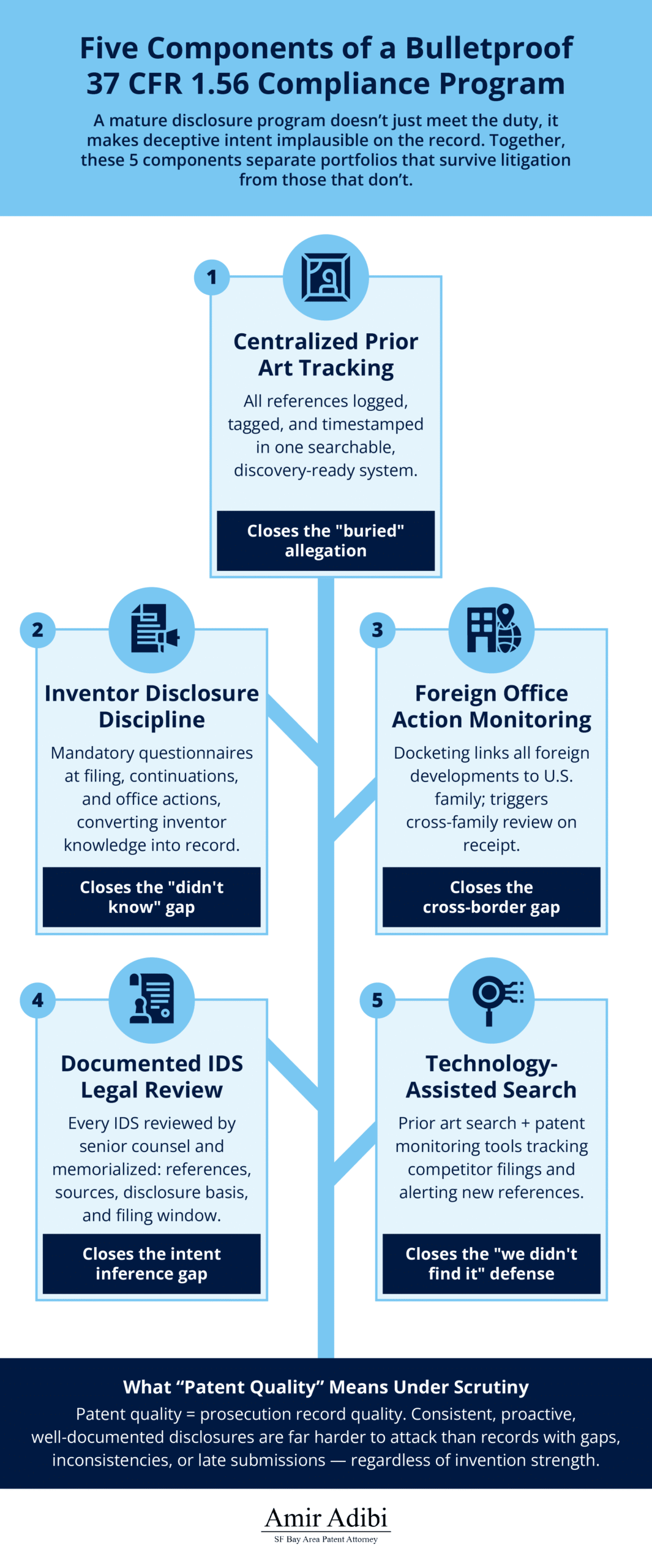
Building a Bulletproof 37 CFR 1.56 Compliance Program
The goal of a mature compliance program is simple to state and hard to build. An inequitable conduct defense should be impossible to raise credibly because the prosecution record and internal workflows make deceptive intent factually implausible. Every disclosure decision has a contemporaneous rationale. Every foreign communication has an audit trail. Every potentially inconsistent regulatory statement has been reviewed by IP counsel before it left the building. The program will not prevent every bad outcome. It will transform the “single most reasonable inference” argument into a losing one.
Designing an Internal Disclosure Workflow That Eliminates “Oversight” as a Defense
An enterprise-level duty of candor program has five operational components. None are individually novel. The discipline is running all five consistently, across every high-value family, for the full life of the prosecution and post-grant record.
Centralized prior art tracking. Every reference that enters the organization’s awareness — from inventors, from foreign associates, from commercial intelligence, from competitor monitoring — is logged in a single system, tagged to the relevant applications, and timestamped at the point of entry. The log is not optional, and it is not a cloud of emails. It is a queryable record that counsel can produce in response to a discovery request and use to rebut allegations that references were buried, ignored, or arrived “too late to disclose.”
Inventor disclosure discipline. Inventors rarely understand that their personal knowledge of competitor products, academic publications, prior commercial use, and their own pre-filing disclosures creates a disclosure obligation. Mandatory inventor disclosure questionnaires — structured to surface each category of potentially material information, administered at filing, and repeated at continuation filing and at response to substantive office actions — convert inventor knowledge into counsel’s knowledge in a way that creates a clean compliance record. The questionnaires should also probe commercial use, sales, public demonstrations, and academic presentations that implicate § 102 bars and inventorship disputes, not just prior art per se.
Systematic foreign office action monitoring. Execute the 7-step protocol described in the global filing section above. The docketing system should surface every foreign development against every co-pending U.S. application in the same family, so that a search report filed in one EPO national phase automatically triggers review of every U.S. continuation that might benefit from the disclosure.
Documented legal review of IDS submissions. Every IDS filing — not just the initial one — should be reviewed by a designated senior attorney, with the review memorialized in a short internal memo identifying the references cited, the source of each reference, the basis for concluding that disclosure satisfies the obligation, and the 37 CFR 1.97 window under which the filing qualifies. That memo is the contemporaneous record that rebuts specific-intent allegations if the patent is later challenged. For high-value families, formal opinion letters on disclosure decisions at key prosecution milestones add another layer of defensibility.
Technology-assisted search. Professional prior art search programs, combined with patent monitoring systems that track competitor filings and generate automatic alerts on new references in technology areas of interest, are increasingly expected infrastructure for sophisticated portfolios.This does not replace the attorney’s disclosure judgment, but closes the “we didn’t know about it” gap that used to be a plausible good-faith defense.
Together, these components produce a compliance posture that a defendant’s expert will struggle to attack. The annual audit that tests the posture is the natural next step, and the checklist below is a defensible baseline for in-house IP strategists running that audit.
The 37 CFR 1.56 Compliance Audit — 12 Questions for an Annual Portfolio Review:
- Does every high-value patent family have a documented chain of custody for foreign search reports and office actions received in the past 12 months?
- Are inventor disclosure questionnaires completed and memorialized at filing, at every continuation filing, and at response to substantive office actions?
- Is every IDS filed in a high-value family accompanied by an internal memorandum identifying sources, filing window, and 37 CFR 1.97(e) certification basis?
- Has the organization identified every individual subject to the duty of candor across every active U.S. application, and confirmed that each has received current training on personal, non-delegable disclosure obligations?
- Are docketing triggers in place to capture every foreign communication within 24 business hours of receipt by foreign associates?
- Has a cross-check been run within the last 12 months to identify references cited in one U.S. application that were not disclosed in co-pending related applications in the same family?
- For pharma and biotech portfolios, is IP counsel integrated into review of regulatory submissions that discuss mechanism of action, comparative efficacy, or prior-art landscape?
- Are EPO opposition proceedings and post-grant challenges on counterpart applications monitored, and are references surfaced in those proceedings evaluated for U.S. disclosure in co-pending applications?
- Is there a documented protocol for handling prior art surfaced in litigation discovery or IPR proceedings — requiring immediate disclosure in every co-pending application and continuation?
- Has the organization audited its active portfolio for shared-inventor, shared-counsel, and overlapping-claim-scope factors that could support an infectious unenforceability finding?
- Are opinion letters documenting good-faith disclosure decisions available for the top 20% of the portfolio by strategic value?
- Has the compliance program itself been pressure-tested by outside counsel experienced in defending inequitable conduct allegations at the Federal Circuit?
Litigation-Proofing Your IDS Strategy: What “Patent Quality” Looks Like Under Adversarial Scrutiny
Patent quality, from a litigation perspective, is not a measure of the invention. It is a measure of the prosecution record. A patent whose file history shows consistent, proactive, well-documented disclosure is exponentially harder to attack on inequitable conduct grounds than one whose file history contains gaps, inconsistencies, or unexplained late submissions — even if both patents cover equally strong inventions.
This changes how sophisticated portfolio managers think about the over-disclosure debate. The contested strategic question in IDS filing is whether to disclose marginally relevant references — the ones that are plausibly cumulative, or plausibly distinguishable, where the disclosure judgment could defensibly go either way. The argument against over-disclosure is real: burying examiners in references of marginal value has historically been criticized by the Federal Circuit, and a sufficiently large IDS can be viewed as an attempt to obscure truly material references in a crowd. The argument for over-disclosure is that the asymmetric risk in high-value families almost always favors submission. If the reference turns out to be immaterial, nothing is lost. If it turns out to be but-for material in litigation, the disclosure defeats the inequitable conduct defense before it is plausibly raised.
The answer is not doctrinal. It is portfolio-specific. For low-value filings in non-core technology areas, aggressive filtering is defensible. For flagship patents, continuation families that will generate licensing revenue, and any patent likely to be asserted in litigation, the risk-asymmetry math almost always favors submission with contemporaneous documentation of why the reference was judged relevant enough to disclose. The examiner’s obligation, under the combined operation of 37 CFR 1.97 and 1.98 as articulated in MPEP § 609, is to consider every item cited in a proper IDS. A well-constructed IDS that walks the examiner through complex prior art, with concise statements of relevance where appropriate, strengthens the patent’s presumption of validity during later litigation. It becomes much harder to argue the USPTO was deceived when the prosecution record shows the examiner was informed, educated, and made a substantive allowance decision with the relevant references in front of them.
The infrastructure is replicable. What separates the portfolios that survive litigation from the ones that don’t is rarely a doctrinal surprise — it is the discipline to build this infrastructure before it is needed.
Why Duty-of-Candor Discipline Decides Which Portfolios Survive Litigation
37 CFR 1.56 compliance is the load-bearing wall of patent enforceability. Treat it as a filing formality and the cost is measured in lost patents and lost portfolios — permanent unenforceability, infectious contamination across the family, and, at the far edge, Walker Process treble damages. The doctrine is not exotic. The case law is not new. The workflows that prevent inequitable conduct findings are not secret. What separates the companies that survive Federal Circuit scrutiny from the ones that lose their crown-jewel patents is operational discipline, applied consistently across the life of every high-value family.
The portfolios that survive litigation are built, not discovered. Build the disclosure infrastructure before the litigation opponent builds the narrative that exploits its absence.
Before your next prosecution milestone, review your duty-of-candor infrastructure with counsel who have seen what fails in litigation. Schedule a portfolio audit or compliance program review →

